|
Q: Why does ice float on water?
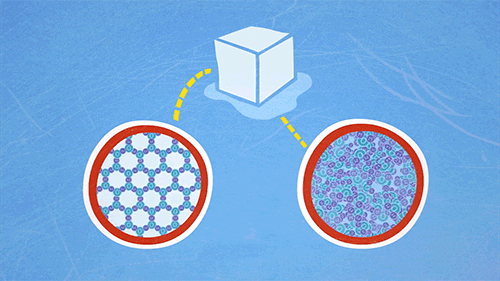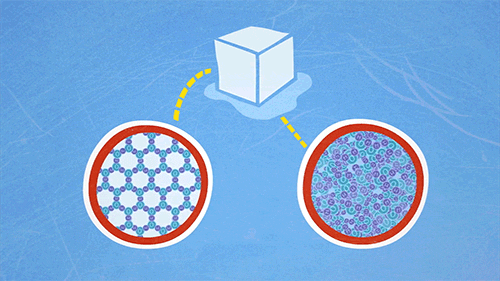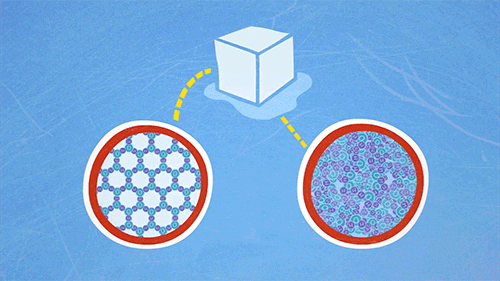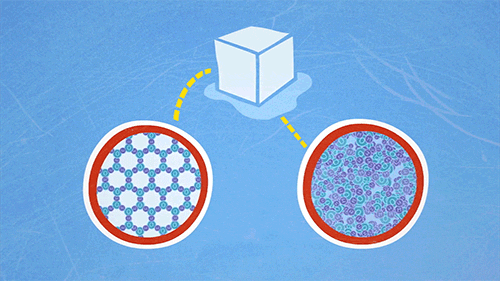
Ice floats on water because it is less dense than water. This may be explained in terms of the arrangement of the H2O molecules in ice and water. In water, the molecules move randomly around each other within the liquid, and are close together. When water freezes into ice, the molecules arrange themselves to form a crystalline lattice (regular arrangement in a repeating pattern) which has more spaces between molecules as compared to water. This increases the volume of ice, which means that ice has a lower density than water, and therefore floats on water.
0 Comments
Q: Why does a less dense object float in a denser liquid? A: Consider the pressure experienced by the object in the liquid. We know two things: 1. Pressure in the fluid = rgh (r = density) 2. Pressure in the fluid is exerted in all directions. The pressure on the upper face of the cube is rgH and acts downwards.
The pressure on the lower face of the cube is rg(H+h) and acts upwards. The net pressure on the cube is rg(H+h) - rgH = rgh. This means that in a fluid, the fluid pressure exerts a net upward force on the cube. The weight of the cube is W = mg. If this force is greater than the weight of the cube, the cube moves upwards in the fluid and floats. However, if this force is smaller than the weight of the cube, the cube will sink to the bottom. |